Week 7 Announcements
Updated October 2nd, 2021
You know the drill, this is your go-to source for information concerning Organic Chemistry class! Past week's announcements will be saved in the Quarter 1 tab. Week 6 Summary
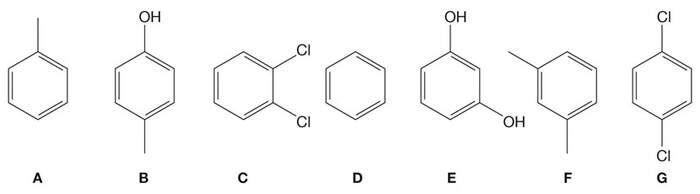
This past week we explored two main topics of interest that are the consequence of intermolecular interactions: (1) Solubility of compounds in water and other solvents (Section 2.7), and (2) ranking relative boiling points of structurally similar compounds. As an example, suppose you were to rank the boiling points of the below compounds from lowest to highest:
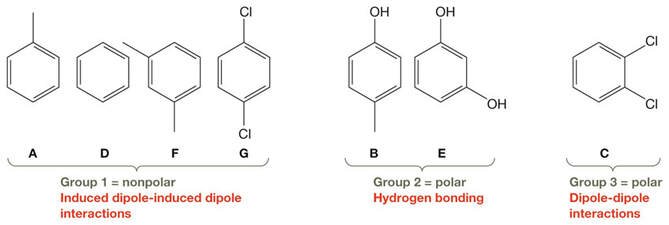
The first step to ranking these is to consider the most significant intermolecular interactions between molecules of each compound, then group these as:
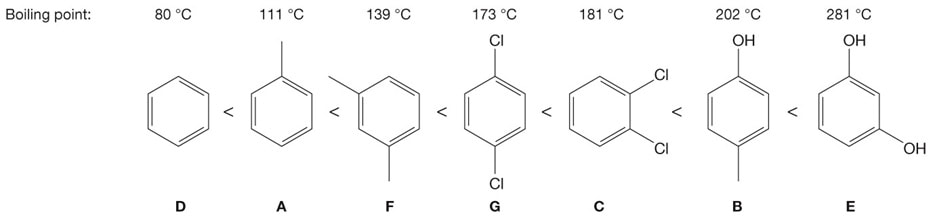
Rank the boiling points within each group. Then combine the array accordingly:
Note that all the above compounds possess a benzene ring, making their structures very similar. It is the substituents attached that provide differences with respect to chemical and physical properties.
We then discussed Section 2.9 - Protic and Aprotic Solvents. Recollect the idea that steric hindrance prevents a solvent like DMSO to solubilize an ionic compound like NaCl. What to expect in Week 7
On Tuesday we will complete Chapter 2, navigating our way through the remaining sections of the text
Introducing... Chapter 3
On Thursday, I will introduce Chapter 3 - Orbital Interactions 1 - Hybridization and Two-Center Molecular Orbitals, which is broken down into the following sections:
So far, we have learned of several different models for describing the shapes of molecules and charge distribution of electrons. While informative, these models still have shortcomings:
|
Week 7 Resources/ Assets
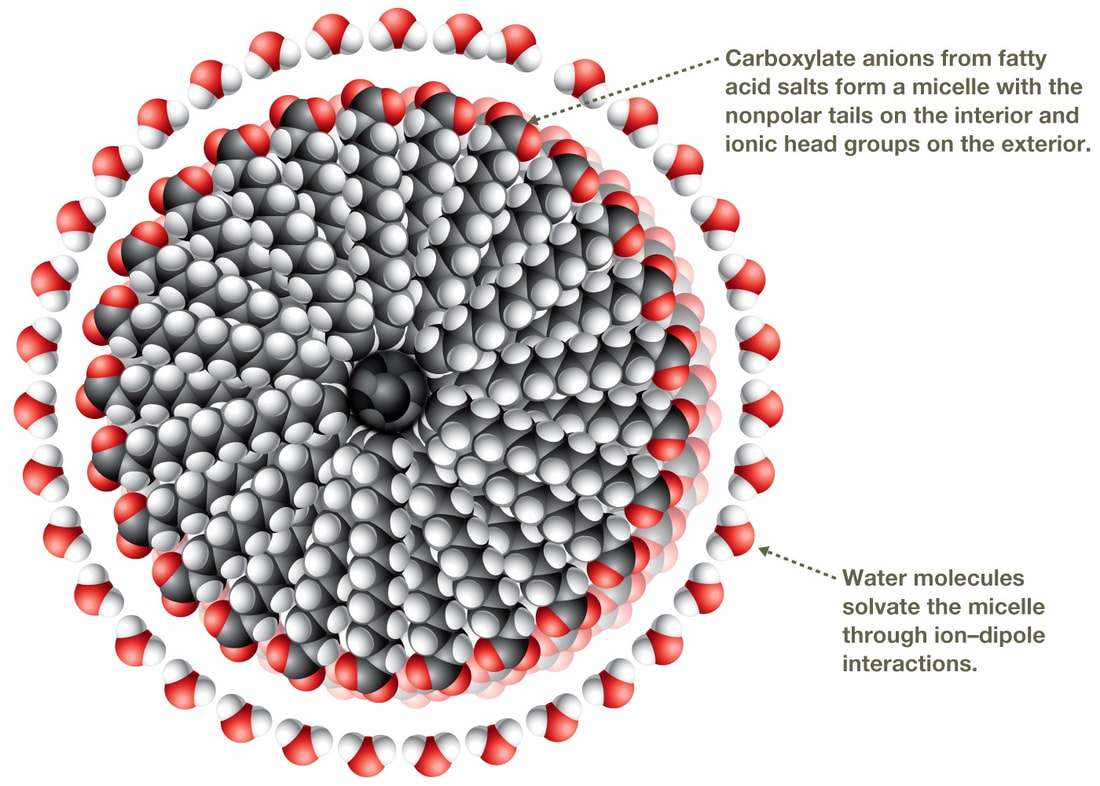
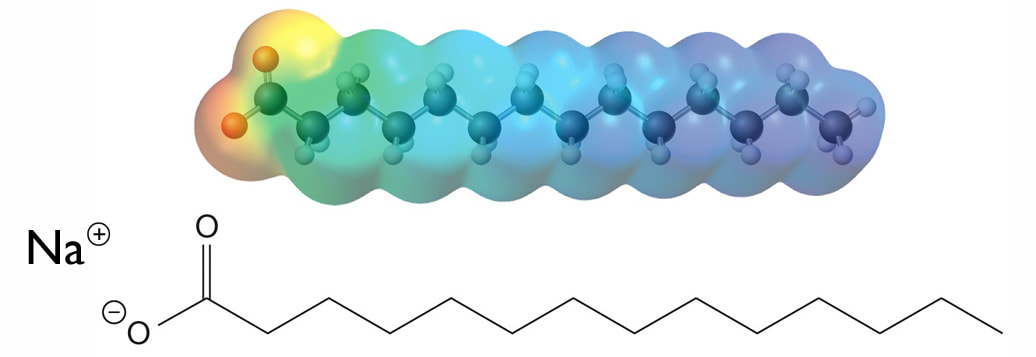
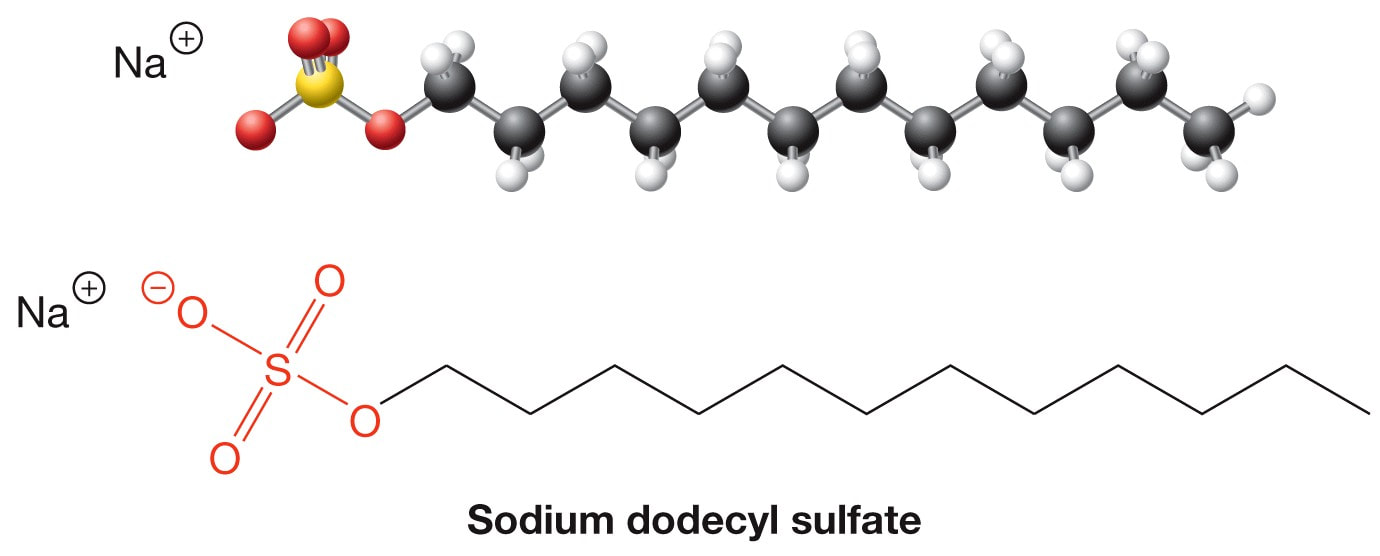
Soaps and Detergents
|